When you start a TNF inhibitor for rheumatoid arthritis, psoriasis, or Crohn’s disease, you’re signing up for powerful relief from inflammation. But beneath that benefit lies a quiet, serious risk: reactivation of latent tuberculosis. This isn’t a rare edge case. It’s a well-documented, life-threatening complication that happens more often than most patients - and even some doctors - realize.
Why TNF Inhibitors Put You at Risk for TB
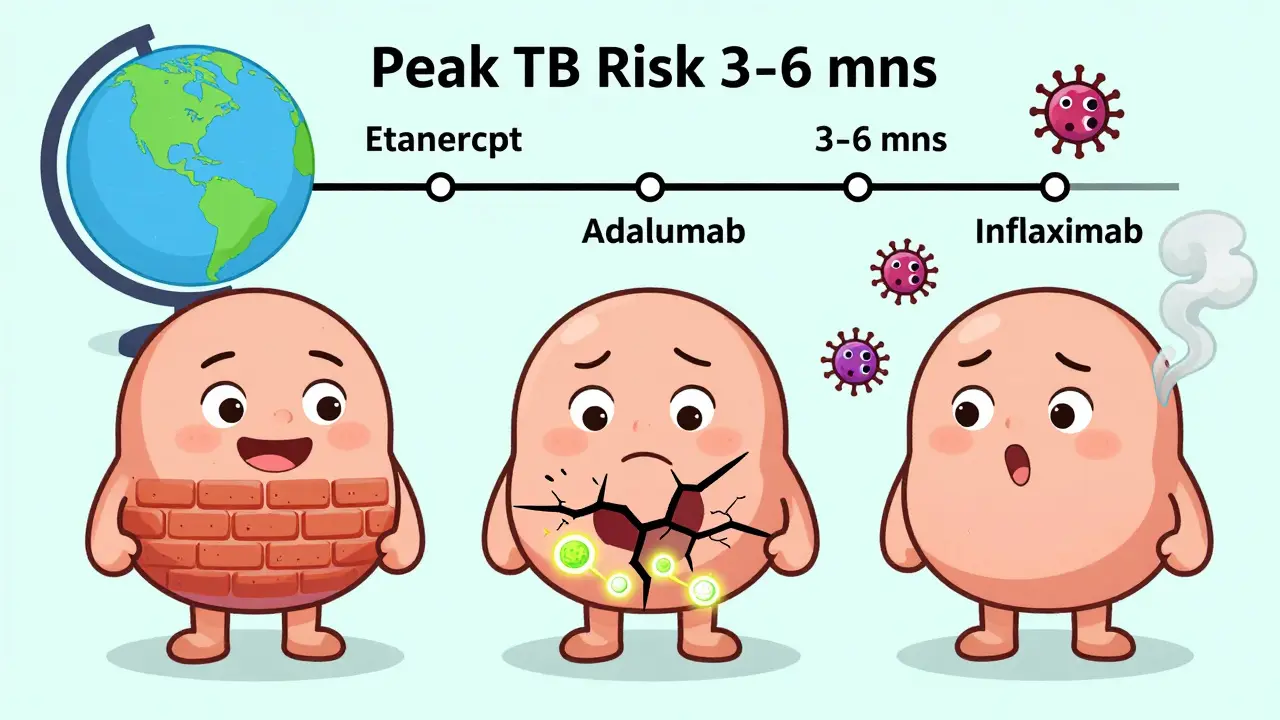
Your body keeps tuberculosis (TB) in check through tiny, tightly packed clusters of immune cells called granulomas. These structures wall off the bacteria, keeping it dormant. TNF-alpha, a protein your immune system makes, is the glue holding those granulomas together. When you take a TNF inhibitor, you’re blocking that protein. And without it, the granulomas fall apart. The bacteria wake up. And then they spread. Not all TNF inhibitors are the same. There are two main types:- Class 1: Etanercept (Enbrel) - This one acts like a decoy receptor. It soaks up excess TNF-alpha without touching the membrane-bound version your body needs to keep granulomas intact.
- Class 2 and 3: Infliximab (Remicade) and Adalimumab (Humira) - These are monoclonal antibodies. They bind tightly to both soluble and membrane-bound TNF. That’s why they’re more effective at shutting down inflammation - and why they’re far more dangerous when it comes to TB.
Screening: What You Must Do Before Starting
Before you get your first TNF inhibitor shot or infusion, you need two things:- A tuberculin skin test (TST) - Also called a PPD test. A small amount of TB protein is injected under your skin. If you’ve been exposed, your immune system reacts with a raised bump.
- An interferon-gamma release assay (IGRA) - A blood test that measures how your immune cells respond to TB proteins. It’s more accurate than TST, especially if you’ve had the BCG vaccine (common outside the U.S.).
What If Your Screening Is Negative?
Here’s the scary part: 18% of TB cases happen in people who tested negative before starting treatment. Why?- False negatives - Especially if you’re on steroids or have HIV, your immune system might not react enough to show up on the test.
- Recent exposure - If you were exposed to TB in the last 8 weeks, your body hasn’t had time to mount a detectable response.
- Test limitations - TST can be affected by BCG vaccination or poor technique.
Monitoring After You Start
Screening isn’t a one-time checkbox. You need to stay alert.- First 3-6 months - This is when most TB cases show up. If you’re on infliximab or adalimumab, you’re at highest risk in this window.
- Check symptoms every 3 months - Fever, night sweats, unexplained weight loss, cough lasting more than 2 weeks. Don’t brush these off as "just a cold."
- Extrapulmonary TB is common - Nearly 80% of TB cases in TNF inhibitor users aren’t in the lungs. They’re in lymph nodes, bones, joints, or even the brain. If you have unexplained joint pain or swollen lymph nodes, get it checked.

What About High-Risk Patients?
If you’re from a country with high TB rates - even if your screening is negative - guidelines now say: treat for latent TB anyway. The European League Against Rheumatism (EULAR) recommends this for anyone from a high-burden region. Why? Because screening tests aren’t perfect. The risk of missing TB is higher than the risk of treating someone who doesn’t need it. And here’s the real-world problem: 32% of patients stop isoniazid because of liver concerns. But the liver damage risk is low - about 1 in 100. And it’s usually reversible. The risk of active TB? Much higher. If you’re worried about side effects, ask about the 4-month combo. It’s safer and easier to stick with.The Bigger Picture
About 2.5 million people worldwide are on TNF inhibitors. That number is growing. And while biosimilars have cut costs - adalimumab now costs $4,500 a month instead of $6,700 - screening adds $150-$300 per patient upfront. In places with limited resources, 80% of clinics can’t even access IGRA testing. That’s why TB reactivation still happens. New drugs are coming. In 2024, early trials of a new class of TNF inhibitors showed an 80% reduction in TB reactivation risk in animal models. These drugs target only soluble TNF, leaving the membrane-bound version alone. That’s the holy grail: control inflammation without risking TB. Until then, the rules are simple:- Screen before you start - with both TST and IGRA if you’re high-risk.
- Treat latent TB before beginning therapy.
- Watch for symptoms - especially in the first 6 months.
- Don’t assume a negative test means zero risk.
Can I still take a TNF inhibitor if I’ve had TB before?
Yes - but only after you’ve completed full treatment for active TB and been cleared by your doctor. You’ll need to wait at least 3 months after finishing TB antibiotics before starting a TNF inhibitor. Even then, you’ll be monitored closely. Your risk of reactivation is higher than someone who’s never had TB.
Is the IGRA test better than the skin test?
For most people, yes. The IGRA (blood test) is more accurate if you’ve had the BCG vaccine (common outside the U.S.) or if you’ve had a previous positive skin test. It doesn’t give false positives from BCG. But it’s not perfect - and it’s not always available. Many clinics still rely on the skin test. The best approach is to use both if you’re at high risk.
What if I’m from a high-TB-burden country but tested negative?
You should still be treated for latent TB before starting a TNF inhibitor. Guidelines from EULAR and the CDC recommend this. Screening tests can miss up to 20% of latent infections, especially in people from high-prevalence areas. The cost and side effects of treatment are far lower than the risk of active TB.
Can I get TB while on a TNF inhibitor even if I’ve been treated for latent TB?
Yes. Treatment for latent TB reduces risk but doesn’t eliminate it. You can still be reinfected, especially if you live in or travel to a high-TB area. That’s why ongoing monitoring - checking for symptoms every few months - is critical, even after treatment.
How long should I wait after treating latent TB before starting a TNF inhibitor?
Most guidelines recommend waiting at least 1 month after starting TB treatment. This gives the antibiotics time to begin working. Some doctors prefer to wait 2-3 months, especially if you’re on a high-risk drug like infliximab or adalimumab. Always follow your doctor’s advice - timing matters.
Are there TNF inhibitors with lower TB risk?
Yes. Etanercept (Enbrel) has the lowest risk - about one-fifth the risk of infliximab or adalimumab. That’s because it doesn’t interfere with membrane-bound TNF, which is critical for keeping TB contained. If TB risk is a major concern for you, etanercept may be the safer choice - but it’s not always the most effective for your condition. Talk to your rheumatologist about balancing effectiveness and safety.






Comments(12)